
Before to start, a few definitions
They are usually hard to measure directly during production but are essential. |
Downstream process comparison between production sites
This This case study compares two downstream processes between two production sites with our HCP-PROFILER Solution. Thanks to this test, you will have an overview of the benefits of an internalized DSP process.
The pharmaceutical industry requires products free from process impurities to meet clinical requirements. This case study compares two downstream processes between two production sites with our HCP-PROFILER solution. The aim of this operation for our customer is to evaluate their current internal processes.
Functioning and strategy
From the cellular system harvest to the final Drug Product, several purification steps are implemented through the Downstream Process (DSP) in order to remove the Host Cell Proteins (HCPs). These residual proteins need to be eliminated because they are part of the Critical Quality Attribute (CQA) since they can affect product:
- Quality
- Stability
- Efficacy
- Safety
- Induce or enhance immunogenicity.
HCPs expression is further influenced by culture conditions that generate differences in matrices contents. Thus, to perform sample comparison in matrices that can be different, it is necessary to get a consistent and reliable analytical method.
Our HCP Profiler Solution is designed to be implemented through the whole DSP steps from harvest to final drug substance. Its repeatability and reproducibility capabilities, provide for each HCP, an identification and a quantification evaluation. It carries out the standard Quality Control tests as low limit of detection evaluation, consistency, and sample prep monitoring. Our solution is provided with dedicated software to check data consistency, enhance data visualization…
→ How does it work?
The functioning of the HCP profiler consists in spiking a universal calibration curve of synthetic peptides accurately calibrated. The calibration curve allows the conversion of the peptide (parts of the proteins) signal into a quantitative measurement of all identified proteins. Peptides used in the calibration curve are chosen by us. All identified proteins are normalized using this calibration curve and the normalized signal is compared batch to batch. The coupling of internal standards with a dedicated software allows the improvement of data process robustness. It facilitates the comparison of independent data from different samples and batches and allows the reproducible quantification of individual HCP.
Thanks to the dedicated software, it is easier to compare protein quantity between different batch production of each HCPs within all production batches. In this example, HCP-PROFILER was used to compare the DSP performances between 2 production sites by monitoring two production batches of a therapeutic antibody. In the will to internalize the production and the purification steps of their drug, our customer needed better knowledge and a deep evaluation of their internal process.
What are the results of the operation?
HCPs identification and quantification permitted to show that the total HCPs content was roughly twice less important with the newly internalized DSP (115 ppm) compared to the externalized one (274 ppm). However, a risky protein, the Phospholipase B-like, was identified in the drug product with both DSPs at similar levels. This protein is known to induce drug product instability through time, probably by degrading adjuvant.
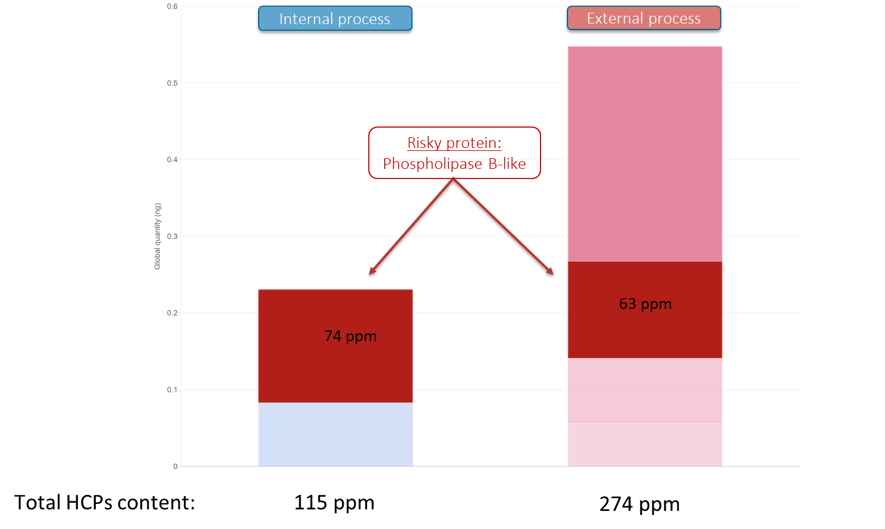
*Figure: Host Cell Proteins (HCPs) quantification comparison between two production sites (internalized or externalized processes)
Thanks to the results, our client was aware of the importance to develop a different and optimized DSP to remove phospholipase protein from the product. Also, the results encouraged us to push forward the internalization of the drug production process and to initiate a scale-up step to achieve more similar operations for our clients.
 Anaquant HCP analysis I Protein characterisation I Protein analysis
Anaquant HCP analysis I Protein characterisation I Protein analysis